Dr Rajiv Desai
An Educational Blog
DENGUE
DENGUE:
Prologue:
I have treated about 150 patients of dengue fever in the year 2008 and 2009 in India and I want to share my experience with the rest of the world about this not-so-bad infamous disease which was wrongly labeled ‘horrible’ as patient’s relatives run from one place to another place in search of platelet transfusion looking at low platelet count with fear and the fear psychosis augmented by treating doctors.
Synonyms and abbreviations:
Dengue fever = DF
Dengue hemorrhagic fever = DHF
Dengue shock syndrome = DSS
Dengue virus = DENV
DIC = disseminated intravascular coagulation
GI tract = gastrointestinal tract
NSAIDS = non-steroidal anti-inflammatory drugs
Normal saline = 0.9 % sodium chloride solution = 9 gram salt dissolved in 1 Liter water.
ICF = intracellular fluid
ECF = extracellular fluid = interstitial fluid + intravascular fluid
Dengue fever is an acute febrile viral illness caused by a bite of an adult female Aedes Aegypti/ Albopictus mosquito (vector/arthropod) transmitting a virus (DENV) from infected mosquito to human. Dengue pronounced as ‘den ghee’ is derived from Swahili phrase ‘ki denga pepo’ meaning ‘cramp like seizure caused by an evil spirit’.
The spread:
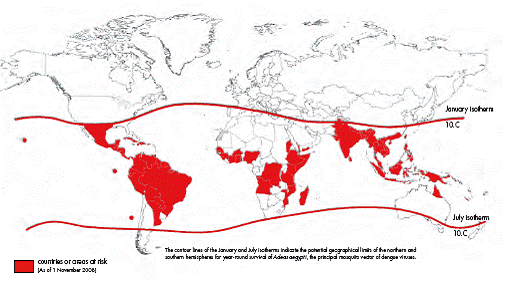
Countries/areas at risk of dengue transmission
Dengue is the most important and most widespread mosquito borne viral infection in humans. There is a dramatic increase in global incidence of dengue in last 20 years. The latest statistics shows that about 2500 million people (two fifths of the world’s population) is at risk of getting dengue fever with annual incidence of about 100 million cases of dengue infections worldwide in more than 100 countries. Only in small proportion of cases, the virus causes bleeding diathesis known as Dengue Hemorrhagic fever (DHF). In 20 to 30 % cases of DHF, a patient develops shock known as Dengue Shock Syndrome (DSS). During epidemics of dengue, infection rates among those who have not been previously exposed to the virus are often 40% to 50%, but can reach 80% to 90%.
H1N1 flu (swine flu) killed 11749 people worldwide in the year 2009 and yet it attracted maximum world attention while DHF caused 500,000 hospitalizations worldwide in the year 2009 with 5 % mortality (25000 deaths) ignored by world. Also, untreated DHF/ DSS are associated with mortality of 20 to 50 %. Also, worldwide, children younger than 15 years comprise 90 % cases of DHF. Also, in a dengue-endemic area, the incidence of dengue infection exceeded 10% in infants. Dengue usually breaks out immediately after the rains in the tropical and subtropical areas of Africa, Asia, Australia, Latin America, the Caribbean basin and the Pacific.
The virus:
Dengue virus (DENV) is a single-strand RNA virus having spherical shape with size 50 nanometer belonging to family Flaviviridae and genus Flavivirus. There are 4 serotypes of DENV from DEN 1 to DEN 4 and all these 4 subtypes are different strains of dengue virus that have 60-80% homology between each other. All 4 viruses have Aedes aegypti mosquitoes as vector and all 4 cause similar illness. Each serotype is sufficiently different so that there is no cross-protection and epidemics caused by multiple serotypes (hyperendemicity) can occur. Recovery from infection by one serotype provides lifelong immunity against that virus but confers only partial and transient protection (few months) against subsequent infection by the other three viruses. There is good evidence to prove that the sequential infection by heterologous serotype increases the risk of developing DHF/DSS.
Transmission:
Dengue viruses are transmitted to humans through the bites of infective female Aedes mosquitoes. Mosquitoes generally acquire the virus while feeding on the blood of an infected person. After virus incubation for 8 to 10 days (extrinsic incubation period), an infected mosquito is capable, during probing and blood feeding, of transmitting the virus for the rest of its life. Infected female mosquitoes may also transmit the virus to their offspring by transovarial (via the eggs) transmission, but the role of this in sustaining transmission of the virus to humans has not yet been defined. However, mosquito eggs can survive up to 6 months at high temperature.
Infected humans are the main carriers and multipliers of the virus, serving as a source of the virus for uninfected mosquitoes. The virus circulates in the blood of infected humans for two to seven days, at approximately the same time that they have a fever; Aedes mosquitoes acquire the virus when they feed on an individual during this period. Aedes mosquitoes are unharmed during infection with dengue virus due to mosquito immunity (molecules of RNA interference) which allows virus to multiply in mosquito without harming it. There is no direct human to human transmission of dengue and hence dengue is not a contagious disease.
When transmission rates are higher, a person stands a better chance of being bitten by more than one strain during this period of cross-immunity. This results in seroconversion and broader immunity, making a person less likely to get a severe dengue infection after they are no longer protected by cross-immunity. But when transmission rates fall, fewer people are able to build a library of antibodies during that year of cross-immunity, leaving them susceptible to subsequent strains down the road. So incidence of DHF/DSS is inversely proportional to dengue transmission rate in a given population in a given year and therefore all efforts must be made to eradicate dengue rather than to have low transmission rate.
Other flavivirus transmitted by mosquito bite are yellow fever virus; Japanese/ St. Louis/ West Nile encephalitis viruses etc.
Pathophysiology:
DHF/DSS usually occurs during a second dengue infection in persons with preexisting actively or passively (maternally) acquired immunity to a heterologous dengue virus serotype. When another subtype of dengue virus infects the individual, the virus will activate the immune system to attack it as if it was the first subtype. The immune system is tricked because the 4 subtypes have very similar surface antigens. The antibodies bind to the surface proteins but do not inactivate the virus. The immune response attracts numerous macrophages, which the virus proceeds to infect because it has not been inactivated. This situation is referred to as Antibody-Dependent Enhancement (ADE) of a viral infection. This makes the viral infection much more acute. The body releases cytokines that cause the endothelial tissue to become permeable which results in hemorrhagic fever and fluid loss from the blood vessels Illness begins abruptly with a minor stage of 2-4 days’ duration followed by rapid deterioration. Increased vascular permeability, bleeding, and possible DIC may be mediated by circulating dengue antigen-antibody complexes, activation of complements and release of vasoactive cytokines. Recent data suggests that endothelial cell activation could mediate plasma leakage from intravascular compartment to interstitial compartment due to transient (24 to 48 hours) and functional effects on post-capillary endothelial cells. Thrombocytopenia (reduced platelet count) is due to decreased platelets synthesis in viral-suppressed bone marrow. Hemorrhage (bleeding) is due to vascular damage (increased capillary fragility), low platelets and DIC.
Normal platelet count is 150,000 to 400,000 per micro-liter (cmm). Patients with DHF have quantitative platelets abnormality in the form of platelet count below 100,000 and also, qualitative platelets dysfunction. The earliest bleeding tendency is diagnosed with the tourniquet (Hess) test. . This test is performed by inflating a blood pressure cuff on the upper arm to midway between diastolic and systolic blood pressures for 5 minutes. The results are considered positive if more than 20 petechiae per square inch are observed on the skin of the arm. The tourniquet test is positive even if the platelet count is above 100,000 because of vascular damage due to immune reaction following DENV infection.
Frank, spontaneous bleeding in skin, gums, nose and GI tract occurs after platelet count has fallen below 10,000 but the count may be higher if qualitative platelets dysfunction exists due to infection itself or use of NSAIDS by doctors for treatment of body pains.
Plasma leak is the leak of plasma proteins from intravascular compartment to interstitial compartment due to transient (24 to 48 hours) increase in vascular permeability due immune mediated enlargement of post-capillary endothelial pores. This protein leak is associated with loss of water from intravascular compartment to interstitial compartment due to osmotic pull of plasma proteins. This is manifested in the patient of DHF/DSS as one or more of the following: greater than 20% rise in average hematocrit level for age and sex, greater than 20% drop in hematocrit level following volume replacement compared to baseline, or signs of plasma leakage (pleural effusion, ascites, hypoproteinemia). The significant and rapid plasma leak can lead to circulatory blood volume depletion resulting in shock known as DSS in which a patient is having restlessness, hypotension, cold clammy skin, rapid weak pulse and narrow pulse pressure. However, it must be remembered that there are many poor patients in developing countries who have severe nutritional anaemia giving lower hematocrit to start with in dengue fever.
The chronology of pathophysiological changes in DF/DHF/DSS is very important for the management of the patient. Thrombocytopenia precedes plasma leak and plasma leak precedes DSS. If there is no thrombocytopenia, there will be no plasma leak and there will be no DSS. The converse is not true. You can have significant thrombocytopenia without DSS as only 20 to 30 % cases of DHF will go into DSS.
Clinical manifestation:
Incubation period is 3 to 7 days after mosquito bite.
Primary dengue infection means that the patient is infected with dengue virus infection of any serotype for the first time and patient was not infected with another flavivirus (yellow fever ) or vaccinated with another flavivirus vaccine and therefore patient does not have any immunity against DENV and so the possibility of DHF/DSS is less. However, if patient had received anti-dengue IgG antibodies from mother during intra-uterine life, even primary dengue infection can produce DHF/DSS.
Secondary dengue infection means the patient is infected with dengue virus infection of heterologous serotype after having had infection with a different serotype DENV in the past or non-dengue flavivirus infection/vaccination. The immune reaction is very robust and may result in DHF/DSS.
Dengue fever (DF) and DHF are distinct conditions; although they are caused by the same virus, and present with the same symptoms in the first few days of the illness, DHF is not just a worsening/complication of dengue fever and poor management of DF does not lead to DHF.
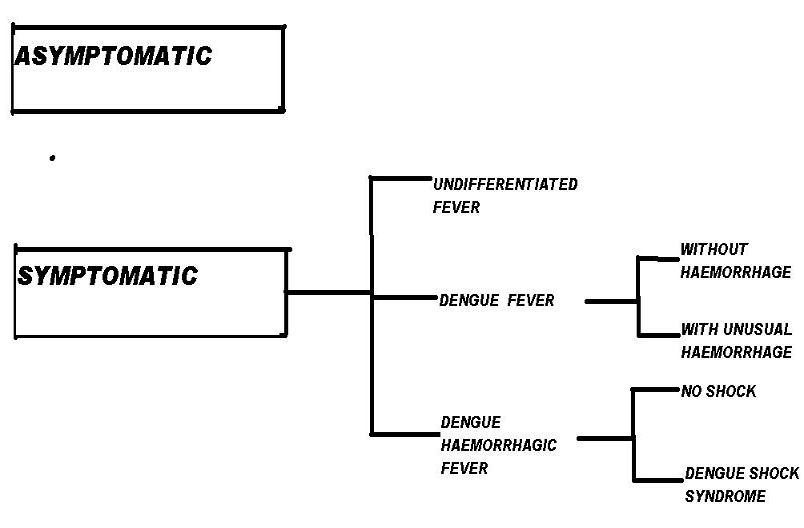
Clinical manifestations of dengue fever
Initial dengue infection may be asymptomatic (50%-90%),may result in a nonspecific febrile illness, or may produce the symptom complex of classic dengue fever (DF). For every patient with symptoms and signs there may be 4-5 persons with no symptoms or with very mild symptoms. After the incubation period, the illness begins abruptly and is followed by the three phases — febrile, critical and recovery.
_
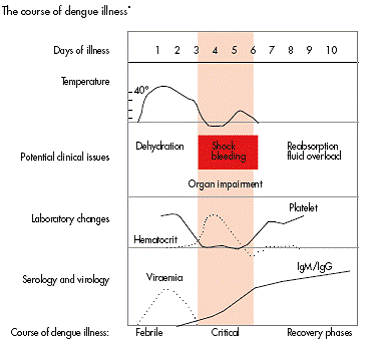
Febrile phase:
Abrupt onset fever ranging from 39.5 to 41.5 degree Celsius accompanied by severe headache, muscle pain (myalgia), joint pain (arthralgia) and retro-orbital pain. Due to severe musculoskeletal pain, dengue fever (DF) is called ‘breakbone fever ‘. Severe bone/ joint/ muscle pain is absent in those dengue patients who are likely to have DHF/DSS. This is associated with anorexia, nausea, vomiting and cutaneous hypersensitivity (skin erythema). The acute febrile phase lasts for 2 to 7 days. There is a maculo-papular skin rash in 50 % cases on trunk spreading centrifugally. In some patients fever subsides in few days and recurs again (saddle shape febrile pattern) with fresh skin rash which is bright red petechiae due to vascular involvement (DHF). The tourniquet test may be positive even with platelet count more than 100,000 per cmm. The WBC count (neutrophils) starts falling from day 1 and the platelet count starts falling from day 3. The platelet count will keep on falling until patient’s temperature returns to normal. Those dengue patients who have DHF start showing bleeding in skin, nose, gums and even GI tract. Women in child bearing age show vaginal bleeding. Liver is often enlarged and tender after few days of fever.
_
_
Critical phase:
This phase starts on day 3 to 7 after onset of illness at the time of defervescence when temperature is returning to normal. There is an increase in capillary permeability associated with plasma leak from intravascular compartment to interstitial compartment which marks the beginning of critical period which lasts for 24 to 48 hours after which plasma leak stops. Leucopenia and thrombocytopenia precedes plasma leak. Although, majority of dengue patients do not develop plasma leak, those who indeed develop worsens with fall in blood pressure, hypoproteinemia and development of ascites or pleural effusion. Hematocrit starts rising as protein leak is accompanied by loss of water from blood making blood thicker with lesser volume.
DSS occurs when critical volume of plasma is lost through leaks rapidly; not giving sufficient time to body’s own corrective mechanism ( Renin- Angiotensin- Aldosterone axis, sympathetic stimulation and ADH release ) to act. Even in nephrotic syndrome, there is protein leak through kidneys but still, body’s corrective mechanism can maintain effective blood volume. Patients with DSS will show lethargy, restlessness, abdominal pain and severe vomiting accompanied by rapid weak pulse, low BP, narrow pulse pressure, cyanosis and cool extremities. Delayed capillary refill may be the first sign of intravascular volume depletion in children. Hypotension usually is a late sign in children. Normal capillary refill time is 2 seconds and a child with DSS may have capillary refill time of 4 to 6 seconds. DSS is fatal if not treated promptly. This plasma leakage is transient lasting for 24 to 48 hours and if circulatory blood volume can be maintained during these critical 48 hours, life can be saved.
Recovery phase:
If the patient survives the 24 to 48 hours of critical phase, then, a gradual reabsorption of extravascular interstitial fluid takes place in 48 to 72 hours. Patient’s general condition and appetite improves, hemodynamic status stabilizes and diuresis ensues. Hematocrit stabilizes/falls followed by rise in WBC count followed by rise in platelet count.
Patient may have respiratory distress due to pleural effusion/ascites during plasma leak period and there-after; and excessive fluid therapy may lead to pulmonary oedema or worsening of pleural effusion/ascites.
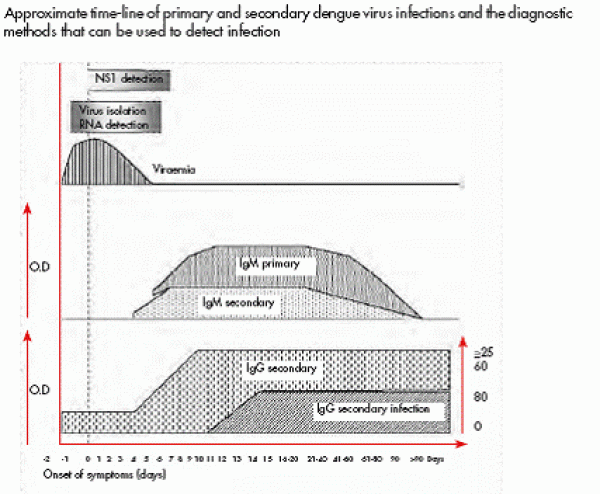
Laboratory diagnosis:
During first 5 days of dengue fever, the DENV and its antigens circulate in blood/serum and therefore DENV can be isolated from blood/serum or RT-PCR test for detection of viral RNA can be done. By detecting dengue NS1 antigen in serum, the Dengue Early Rapid test is able to provide a specific diagnosis of dengue infection in 15 minutes even on the first day of fever. After 5 days, virus and its antigens disappear from blood and the antibodies appear depending on whether it is a primary dengue infection or secondary dengue infection. Both IgM and IgG anti-dengue antibodies neutralize virus which can be detected by ELISA test. During primary dengue, IgM appears after 5 days and peaks after 2 weeks from onset of illness and then declines to undetectable level in 2 to 3 months. IgG also appears in primary infection after 7 days but in very low level but increases slowly thereafter in several months and may remain elevated life time. . In secondary (anamnestic) dengue infection which may result in DHF/DSS, the predominant antibodies are IgG which appear after 5 days in higher concentration and remain high titer for 10 months to life. The IgM also appear in secondary dengue infection but in low level as compared to primary infection and may be undetectable in some cases. The presence of DENV confirmed by RT-PCR test and anti-dengue IgG antibodies simultaneously in blood/serum proves secondary dengue infection. So if a patient suspected of dengue has IgM positive and IgG negative after 5 days of illness (primary dengue), the prognosis is good but if the patient has IgG strongly positive and IgM positive/negative after 5 days of illness (secondary dengue), then, patient may have DHF/DSS. So anti-dengue antibodies tests not only help in diagnosis but also in prognosis of a patient provided it is done at appropriate time. However, false positive test can occur if a person was infected with another flavivirus or received flavivirus vaccine.
DF has leucopenia, normal hematocrit and normal or sometimes low platelet count.
DHF has platelet count less than 100,000 per cmm and hematocrit rise more than 20 % of base value.
Platelet count starts falling after third day of illness and therefore normal platelet count in first three days of illness does not rule out DHF.
Every suspected dengue patient must have at least two complete blood counts ( platelets + hematocrit) at the interval of 24 hours starting from day 3 onwards to judge the fall in platelets with rise in hematocrit to detect DHF at the earliest to prevent DSS.
Patient of dengue may have abnormal liver function tests or abnormal coagulation profile or lower complement C3 level. X-ray chest may reveal pleural effusion and USG abdomen may reveal ascites whenever there is significant plasma leak.
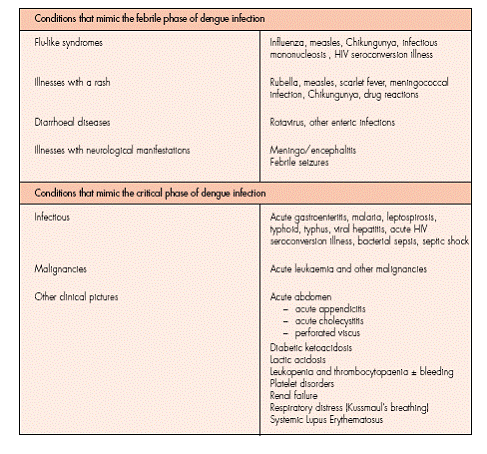
Differential diagnosis:
Iatrogenic complications (caused by wrong management) include sepsis, pneumonia, overhydration and wound infection.
Treatment:
DF treatment includes rest, hydration, paracetamol (acetaminophen) and avoidance of other NSAIDS. Encourage oral intake of oral rehydration solution (ORS), fruit juices and other fluids containing electrolytes and sugar to replace losses due to fever and vomiting. Paracetamol and tepid sponging are used to control fever. NSAIDS other than paracetamol (for example aspirin, ibuprofen, diclofenac, nimesulide, naproxyn etc) are contraindicated as they all affect platelet function, promote bleeding and may cause gastritis. Antibiotics are not indicated.
The patient should be admitted in hospital if any of the following occur:
no clinical improvement, hemorrhage (bleeding in skin, mouth, nose, black stools, coffee-ground vomiting etc), deterioration around the time of defervescence, severe abdominal pain, persistent vomiting, cold extremities, lethargy, restlessness, not passing urine for more than 6 hours, platelet count less than 20,000 per cmm, hematocrit rising more than 20 % of base value, coexisting conditions which may make dengue management complicated (pregnancy, infancy, old age, obesity, diabetes mellitus, renal failure, hemolysis) etc.
In order to understand scientific basis and rationale behind DHF/DSS treatment, please study carefully dengue chronology and physiology of normal fluid ballance.
PHYSIOLOGY OF NORMAL FLUID BALANCE
Treatment of DHF without DSS:
I want to emphasize the point that only 20 to 30 % cases of DHF go into DSS (DHF grade III and grade IV are known as DSS). So 70 to 80 % cases of DHF will always be in grade I and grade II with rare mortality. It is very unlikely that DHF grade II having spontaneous bleeding in skin/gums/nose die due to hemorrhagic shock. Avoid insertion of nasogastric tube to detect concealed bleeding as it is hazardous. Most patients of DHF grade I and II do not need blood transfusion except when bleeding in GI tract or pre-exiting anaemia. The issue is the panic reaction initiated by treating doctor among patients looking at low platelet count without any bleeding. I have seen many patients suspected of dengue received platelet transfusion having platelet count in the range of 30,000 to 60,000 per cmm without any bleeding. This practice must be strongly discouraged. I myself have treated hundreds of dengue patients having platelet count between 30,000 to 60,000 without any platelet transfusion; successfully without any morbidity/ mortality as all of them did not have any spontaneous bleeding due to lower platelet count. The treating doctor and the patient must not fall prey to the number game of platelet count. I recommend platelet transfusion only if a patient has spontaneous bleeding tendency with any platelet count or platelet count less than 10,000 per cmm without spontaneous bleeding tendency. I also recommend platelet transfusion with platelet count between 10,000 to 20,000 per cmm if patient had inadvertently received NSAIDS, pre-existing co-morbidities, difficult to get platelet count repeatedly done etc. Volume replacement for DHF grade I and II, is to initiate IV therapy with normal saline (vide infra) at the rate of 6 ml/kg/hr for 1 to 2 hours and then adjust drip rate as per clinical condition and discontinue IV after 24 to 48 hours. Maintenance fluid requirement should be calculated from Halliday & Segar formula.
Prevention/ treatment of DSS:
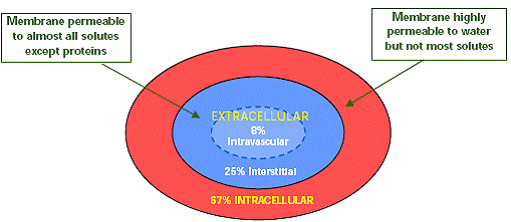
60 % of body weight in men and 50 % of body weight in women is water.
Total body water is distributed between 3 fluid compartments in body.
Intracellular water (intracellular fluid-ICF) makes up about two- thirds of total body water, with remaining one-third, the extracellular water (extracellular fluid- ECF) being distributed between intravascular (25 %) and interstitial (75 %) compartment. So only 8 % of total body water (fluid) contributes to circulating blood volume with RBC (hematocrit) contributing the remaining part of the circulating blood volume.
The cell membrane separates ICF from ECF. The cell membrane is highly permeable to water but impermeable to most solutes and proteins. The endothelial cells of capillary separates intravascular compartment from interstitial compartment. The pores between endothelial cells in capillary allow free movement of water and solutes but do not allow proteins to pass through.
So water can travel in all 3 fluid compartments freely depending on osmolarity of the fluids. Solutes can travel only between intravascular compartment and interstitial compartment and not to ICF barring few exceptions. Colloids can not travel anywhere from intravascular compartment as endothelial pores can not allow its passage. When hypotonic solution 5 % dextrose is given intravenous (intravascular) which is free of sodium, it is as good as free water because in a non-diabetic person, dextrose (glucose) is rapidly transported in the cells and has very little persistent osmotic effect and therefore free water will rapidly travel in all 3 fluid compartments to maintain osmolarity. Out of l liter 5 % dextrose, only 80 ml will remain in intravascular compartment in minutes and therefore hypotonic solutions are contraindicated in shock where the priority is to maintain intravascular blood volume. Also, rapid infusion of hypotonic solution may lead to hemolysis and brain oedema. When normal saline (0.9 % NaCl) is given intravenous, it will be retained only in ECF as free water is not available to travel into ICF and 1 liter of normal saline will give 250 ml fluid in intravascular compartment which will help maintain circulatory blood volume and remaining 750 ml will travel into interstitial compartment. When colloid solutions (dextran, starch, gelatin, albumin) is given intravenous, the entire colloid will remain intravascular as endothelial pores can not pass large colloid molecules and this intravascular colloid will increase osmolarity of blood which will draw water from interstitial compartment into intravascular compartment increasing circulating blood volume much more than the volume of colloid solution. Hence, theoretically, in normal humans, colloid solution is better than crystalloid solution (normal saline or ringer lactate) as far as increasing circulating blood volume is concerned.
During DSS, there is a transient (24 to 48 hours) leak of plasma proteins through post-capillary endothelial pores from intravascular into interstitial compartment accompanied by free water which rushes into interstitial compartment to maintain osmolarity which in turn causes depletion of circulatory blood volume and shock. This shift of water from blood increases hematocrit as RBC volume is relatively greater than plasma volume.
‘It must be remembered that fall in platelet count precedes rise in hematocrit and rise in hematocrit precedes DSS and therefore frequent monitoring of platelet count and hematocrit can detect cases which are going in DSS.’
So best way to prevent DSS is to give plenty of suitable intravenous fluid to a patient who has shown rising hematocrit and who is not in shock as such. Also, the only known effective treatment in DSS is timely, aggressivefluid resuscitation.
So which is the best fluid for prevention and early treatment of DSS?
Various studies have shown that both colloids and crystalloids are equally effective with colloid scoring over crystalloid in selected patients with severe shock having very low pulse pressure.
I humbly disagree and request more studies.
When intravenous colloid (dextran, starch, albumin, gelatin) are given to treat DSS, these large colloid molecules are not retained intravascular as endothelial pores which allow plasma leak in the first place to initiate DSS, also allow colloid molecules to leak out into interstitial compartment dragging free water along with it and therefore the contribution of these colloid solutions to increase circulating blood volume is reduced. Also, colloid solutions can cause allergic reaction and affect coagulation parameters.
On the contrary, 25 % of the infused normal saline will be retained intravascular to increase circulatory blood volume to overcome shock; at the same time without allergic reaction and without affecting coagulation profile. Of course, normal saline is cheaper than colloid solutions and the cost is very important in developing countries. So there is overwhelming evidence to use normal saline over colloid solutions. I myself have used normal saline to treat plenty of DHF patients going into DSS and almost all lives were saved. I could not use colloid solutions to poor patients as it is expensive but in the hindsight, poverty was blessing in disguise as normal saline saved their lives.
I also recommend against using Ringer lactate solution for DSS patients as almost all patients have persistent vomiting with loss of stomach acid giving rise to hypochloraemia & metabolic alkalosis and the lactate anion provides an alkalizing effect resulting from simultaneous removal by the liver of lactate and hydrogen ions and therefore you can not give alternate bicarbonate source to a patient having alkalosis. Also, Ringer lactate should be avoided in liver failure as lactate will accumulate in blood and many patients of DSS have hepatomegaly with abnormal liver function. In fact, normal saline with pH 5 will help alleviate metabolic alkalosis due to vomiting and also replace chloride. Also, normal plasma osmolarity is 285 to 300 mOsm/L and osmolarity of normal saline is 308 and osmolarity of ringer lactate is 274. So normal saline will perform better than ringer lactate to elevate plasma osmolarity in a patient having DHF/DSS who has lower plasma osmolarity due to protein leak. It must be emphasized that higher is the plasma osmolarity, greater is the ability of plasma to pull water from interstitial compartment into intravascular compartment to maintain circulatory blood volume.
Occasionally, a DHF/DSS patient may have only diarrhoea with bicarbonate loss where you can use ringer lactate solution to treat a patient.
My volume replacement recommendations for DHF/DSS are different from WHO.
WHO recommends normal saline, ringer lactate and colloid solutions for treatment of DHF/DSS. In the past, WHO had also recommended hypotonic solution half-strength saline.
I recommend only normal saline for treatment of DHF/DSS and I am confident that results with only normal saline will be better than any other fluid provided large double-blinded trial are conducted.
Volume replacement for DHF grade III and IV (DSS) is to initiate IV therapy with normal saline at the rate of 10 ml/kg/hr for 1 to 2 hours and then adjust drip rate as per clinical condition and discontinue IV therapy after 24 to 48 hours. Maintenance fluid requirement should be calculated from Halliday & Segar formula.
There is no therapeutic difference between normal saline (NS) and 5 % dextrose in normal saline (DNS) as far as restoration of circulatory blood volume is concerned but rapid infusion of DNS may lead to hypokalemia as rapid dextrose entry stimulates insulin which will push potassium in the cells. However, if a patient is persistently vomiting and not able to retain food, then, IV dextrose (glucose) is needed to prevent protein catabolism & starvation ketoacidosis and under such circumstance, DNS may be used alternately with NS.
Corticosteroids, dopamine, dobutamine, noradrenalin etc are of no use in DHF/DSS.
Oxygen must be given to all patients of DSS. Electrolyte, acid-base and glucose imbalances are common in severe dengue and must be corrected.
It must be noted that plasma leak and DSS can occur without frank bleeding and it is not uncommon. I have seen many dengue patients who have low platelet count without bleeding and going into shock state. So absence of bleeding does not mean that the patient is not seriously ill. Also, dengue fever (DF) per se can have hemorrhages due to increased capillary fragility with normal platelet count and patient is not seriously ill. In fact, the term dengue hemorrhagic fever (DHF) is misleading as undue emphasis is given to bleeding and the better term would be “dengue plasma leak fever”. Volume replacement rather than platelet transfusion is the key to save life.
Patient of DHF/DSS may show fall in hematocrit either due to volume replacement or due to bleeding in GI tract. Patient will show clinical improvement if fall in hematocrit is due to volume replacement but patient will deteriorate clinically if hematocrit fall is due to bleeding.
Appropriate management of severe dengue can reduce case-fatality rate to less than 1 %.
There is no vaccine available to prevent dengue till date.
There is no anti-viral drug available for dengue.
Aedes mosquito control measures and prevention of mosquito bite is the only way to prevent dengue fever which I have already discussed on the topic of Chikungunya. Good hygiene and good sanitation in households and its surrounding can prevent mosquito breeding. Global warming has allowed the mosquitoes, and hence dengue fever, to spread to areas where the disease had not previously been known. Drought conditions in some areas also have worsened the outbreak because people have stored water in and near their living areas, creating breeding grounds for mosquitoes that harbor the virus. Floods may indirectly lead to an increase in vector-borne diseases through the expansion in the number and range of vector habitats. Standing water caused by heavy rainfall or overflow of rivers can act as breeding sites for mosquitoes, and therefore enhance the potential for exposure of the disaster-affected population and emergency workers to dengue. Flooding may initially flush out mosquito breeding, but it comes back when the waters recede.
Out of all dengue cases worldwide, less than 1 % are severe dengue cases and out of all severe dengue cases, less than 1 % die provided adequate treatment is given in time.
THE MORAL OF THE STORY:
1) WHO recommends misleading terminology and unscientific treatment for severe dengue.
2) Best way to prevent death in a patient of dengue is to identify which patient is likely to have DHF/DSS by monitoring platelet count and hematocrit from third day onwards and prevent/treat DSS by administrating normal saline (0.9 % NaCl) intravenously to maintain circulatory blood volume. Volume replacement with fluids other than normal saline is not recommended.
3) Platelet transfusions are unnecessary in most cases of severe dengue. Also, many dengue cases worsen due to overzealous treatment rather than severity of illness.
Dr.Rajiv Desai. MD.
April 1, 2010
Postscript:
This is my view on dengue and it is not a gospel truth. Anybody in the world has a right to disagree with my view but the disagreement must be based on facts and logic rather than cynicism.
__________________
Dengue Update 2014:
_
Dengue Epidemic:
_
This year in 2014, till today, I have seen and treated about 70 cases of dengue fever in last 2 months in a small town Daman where I live. Every hospital in Daman and nearby town Vapi has plenty of cases of dengue fever. Many villages surrounding Daman and Vapi have cluster of dengue cases. Some families have 2 to 3 dengue cases in their family. Sadly, media coverage of dengue fever is zero. Sadly, mosquito control measures have been inadequate. Nonetheless, I want to save lives and that is why I am posting this comment.
_
Please remember following points while dealing with dengue fever:
Point-1:
There is no specific symptom or sign of dengue fever. High index of suspicion and cluster of fever cases may lead you to dengue fever.
Point-2:
Because antibodies are detected later, RT–PCR has become a primary tool to detect virus early in the course of illness. Current tests are between 80-90% sensitive, and more that 95% specific. A positive PCR result is a definite proof of current infection and it usually confirms the infecting serotype as well. However, early diagnostic approaches, including virus isolation and molecular tests such as reverse transcription PCR, are still expensive, unavailable and need expertise to perform.
Point-3:
The non-structural protein 1 (NS1) of the dengue viral genome has been shown to be useful as a tool for the diagnosis of acute dengue infections. Dengue NS1 antigen has been detected in the serum of DENV infected patients as early as 1 day post onset of symptoms (DPO), and up to 18 DPO. The NS1 ELISA based antigen assay is commercially available for DENV and many investigators have evaluated this assay for sensitivity and specificity. It allows rapid detection on the first day of fever, before antibodies appear some 5 or more days later. A study found that NS1 Ag positivity varied from 71.42% to 28.4% in acute and early convalescent sera. I have seen few cases of classical dengue with NS1 Ag test negative.
Point-4:
A study found that 80% of all dengue cases have detectable IgM antibody by day five of illness, and 93% of cases have detectable IgM by day six to ten of illness, which may then remain detectable for over 90 days. The combination of NS1 and IgM assay would further enhance the sensitivity of the tests. I have seen few classical dengue cases with both NS1 and IgM test negative. One limitation of IgM testing is cross reactivity with other flaviviruses including West Nile virus (WNV), St. Louis encephalitis virus (SLE), Japanese encephalitis virus (JEV) and yellow fever virus (YFV).
Point-5:
Samples with a negative IgG in the acute phase and a positive IgG in the convalescent phase of the infection are primary dengue infections. Samples with a positive IgG in the acute phase and a 4 fold rise in IgG titer in the convalescent phase (with at least a 7 day interval between the two samples) is a secondary dengue infection. This is important as dengue shock syndrome and death occurs essentially in secondary dengue infections. It is not the virulence of virus that kills human but human’s exaggerated immune response lead to plasma leak and shock.
Point-6:
Normal platelet count in first few days of fever does not rule out dengue fever.
Point-7:
Dengue fever can occur with normal platelet count throughout illness. However, such patients are unlikely to die as plasma leak accompanies or follows fall in platelet count. I have seen a patient who had normal platelet count for 5 days and then suddenly developed fall in count with plasma leak (ascites) and the patient went home well. It takes few weeks for platelet count to return to normal.
Point-8:
The sine qua non of severe dengue is fall in platelet count accompanied or followed by rise in hematocrit (PCV). This is more sensitive and more specific than NS1 Ag and IgM test. All you need is two CBC 24 hours apart. Very rarely you can have other viral infections or typhoid fever or malaria with low platelet count who get dehydrated due to persistent vomiting raising PCV mimicking severe dengue.
Point-9:
Intravenous normal saline in adequate dose at appropriate time is the gold standard of treatment of severe dengue. All you need is to give IV 0.9 % saline 3 to 4 liters (for adults) in 24 hours for 2 to 4 days to save life. The plasma leak is transient. The elevated PCV will become normal in 2 to 4 days. 25 % of the infused normal saline is retained intravascular to increase circulatory blood volume to overcome shock; at the same time without allergic reaction and without affecting coagulation profile. Also, normal saline elevates plasma osmolarity in severe dengue who has lower plasma osmolarity due to protein leak. Higher the plasma osmolarity, greater is the ability of plasma to pull water from interstitial compartment into intravascular compartment to maintain circulatory blood volume. This is how normal saline prevents and/or treats plasma leak and maintains circulatory blood volume to prevent and/or treat dengue shock syndrome. Nobody dies due to low platelet count but low blood pressure due to low circulatory blood volume due to plasma leak causes multiple organ failure. All my 70 patients received IV normal saline. All went home. There is a possibility of overload with normal saline. Overloading may occasionally give rise to puffiness of face or edema feet or pulmonary congestion but it will revert back to normal in due course of time. Avoid diuretic use to remove excess fluid unless you are sure that plasma leak has sealed because diuretics will cause intravascular volume depletion and worsen or precipitate shock. I have never experienced problem of volume overload as normal heart and normal kidneys can take care of excess fluid.
Point-10:
If anybody in the world want to do double blind randomized controlled trials of dengue patients with normal saline versus any other fluid, please go ahead. If I am proved wrong, I will quit medical practice and apologize to world. In 1999, a Randomized Double-Blind Comparison of 4 Intravenous Fluid Regimens (dextran, gelatin, lactated Ringer’s, and “normal” saline) for initial resuscitation of 230 Vietnamese children with dengue shock syndrome were conducted. All the children survived, and there was no clear advantage to using any of the 4 fluids. However, in 2012, Cochrane systematic review evaluated the effects of colloids versus crystalloids in all critically ill patients included 74 trials, 66 of which reported mortality data. The review found no evidence that resuscitation with colloids reduced the risk of death compared to crystalloids. WHO in 2012, changed its recommendation (of 2009) for dengue fever volume replacement; and stated that there is lack of effectiveness of colloids compared to crystalloids in critically ill patients and due to higher cost of colloids, there is no justification for the inclusion of colloids for volume replacement in Dengue patients in the WHO List of Essential Medicines.
Point-11:
Antibiotics and corticosteroids have no role in dengue fever. Rarely if patient develops secondary bacterial infection with high neutrophil count, you may add antibiotics.
Point-12:
Severe abdominal pain with fever could be beginning of plasma leak, dengue ascites and dengue shock syndrome. Severe abdominal pain of dengue fever mimics appendicitis, salpingitis and cholecystitis.
Point-13:
The primary preventative measure to reduce dengue infections is the control of mosquito populations. Because the transmission of dengue requires mosquitoes as vectors, the spread of dengue can be limited by reducing mosquito populations. The average lifespan of an Aedes mosquito in nature is two weeks. However, since the dengue virus can be passed from adult to egg, the dengue virus is guaranteed to survive until the next summer and heavy rains. The eggs can lie dormant in dry conditions for up to about nine months, after which they can hatch if exposed to favorable conditions, i.e. water and food. Therefore in my view, destruction of mosquito eggs is the best way to prevent dengue and not killing mosquitoes alone.
________
Dr. Rajiv Desai. MD.
September 25, 2014
__________
Unremitting dengue epidemic:
Dengue epidemic in small town Daman and surrounding villages is unremitting as I post this comment today. I have seen and treated additional 50 patients of confirmed dengue fever cases, so total number of dengue patients treated by me this season adds up to 120 till today. Some additional points to be discussed:
Point-1:
Unwarranted horror of low platelet count:
Patients and relatives are panicked by seeing low platelet count. Just as fever is a symptom of infection although non-infectious causes can also cause fever, low platelet count (thrombocytopenia) is a sign of dengue fever although low platelet count can occur in many conditions including alcoholism, drugs, vitamin B-12 deficiency, malaria, typhoid, other viral infections, sepsis and immune disorders. Fever does not kill you unless fever becomes disease itself e.g. hyperpyrexia (core body temperature > 41.5 °C [106.7 °F]); in the same way, low platelet count does not kill you unless low platelet count causes bleeding. However, I have seen many dengue patients with platelet count around 10,000 to 20,000 per microliter of blood who do not bleed. WHO says that prophylactic platelet transfusions for low platelet count in otherwise hemodynamically stable patients have not been shown to be effective and are not necessary. I recommend platelet transfusion [Single Donor Platelet Concentrate (SDPC)] only if patient has bleeding tendency with low platelet count or platelet count less than 10,000 per micro-liter of blood without any bleeding tendency. Death in dengue occurs due to irreversible shock due to plasma leak. Irreversible shock itself causes multiple organ failure and disseminated intravascular coagulation (DIC), and DIC itself can cause bleeding. So bleeding in dying dengue patient is not unusual. However in acute DIC, PT and aPTT are prolonged, platelet count & fibrinogen decrease and D-dimer, FDP, and fibrin monomer levels are elevated. The essence of dengue management is maintenance of circulatory blood volume and not platelet count.
Point-2:
Unpredictability of severity of dengue:
Top Mumbai doctors say that 90 % dengue patients do not need admission in hospital. I humbly disagree. After seeing hundreds of dengue patients, I assert that dengue fever is an unpredictable illness. You may have platelet count 200,000 with fever and next day it may become 50,000 with plasma leak without fever. Even though secondary dengue infection leads to plasma leak, I have seen few dengue patients with primary infection getting plasma leak. This is because immune response to dengue virus determines plasma leak and occasionally primary dengue infection can lead to severe immune reaction and plasma leak. Also person may have been infected in past with other viruses similar to dengue virus and therefore primary dengue infection leads to exaggerated immune response. From clinical examination and blood tests, nobody in the world can predict that this dengue patient will not develop plasma leak and shock. Yes, rising hematocrit (PCV) and falling platelet count does predict adverse outcome but it can happen suddenly in otherwise stable patient. Therefore I recommend hospitalization of every confirmed dengue fever case to monitor platelet count, PCV and blood pressure. Patient should not be discharged from hospital as soon as fever subsides because plasma leak often develops when fever is subsiding. For those patients with plasma leak, discharge is to be deferred till PCV start falling and patient start eating food. Asymptomatic low platelet count is not a criterion for admission, management and discharge of patient but should be seen as a sign of dengue fever although dengue fever can occur with normal platelet count and low platelet count can occur in many illnesses besides dengue.
__________
Dr. Rajiv Desai. MD.
October 15, 2014
________________
Dengue Virus Mutation:
Dengue virus is an RNA virus and mutations are a way of life for an RNA virus, and mutations come and go every time a genome replicates – it is likely that every single genome copy of an RNA virus has a mutation. Longer the dengue epidemic lasts; greater will be the mutations of dengue virus. Viral mutations are evolutionary biologically hardwired to confer greater reproductive fitness to virus. The key is to determine whether these mutations affect any of the biological properties of the virus, such as transmission, stability, or virulence. I have seen many dengue patients present as upper respiratory tract infection (sore throat, cough and cold). I don’t know whether the dengue virus will spread from one person to another by droplet infection rather than by usual mosquito vector transmission. Other doctors have seen many dengue patients presenting as gastritis. Some doctors have seen dengue patients getting jaundice with liver necrosis. All these findings suggest mutation of dengue virus and virus is mutating because health authorities are unable to control dengue epidemic rapidly. Remember, longer the dengue epidemic lasts, stronger the dengue virus becomes.
Dr. Rajiv Desai. MD.
21 November 2014
______________



Wonderful goods from you, man. I’ve keep in mind your stuff previous to and you’re simply extremely great. I actually like what you have obtained right here, really like what you are saying and the way during which you are saying it. You are making it entertaining and you still care for to keep it wise. I can’t wait to learn far more from you. This is really a great web site.
I would like to show my admiration for your generosity for people that have the need for help on this concern. Your real commitment to passing the solution around turned out to be rather helpful and has in every case allowed people just like me to realize their targets. Your personal important facts implies a lot a person like me and a whole lot more to my office colleagues. Thank you; from all of us.
Great website! I am loving it!! Will come back again. I am bookmarking your feeds also.
Good luck to you and your work, good friend. It is really pleasing to see such posts these days when knowledge and sharing have lost their importance.
Good luck to you and your work, good friend. It is really pleasing to see such posts these days when knowledge and sharing have lost their importance.
You need to be a part of a contest for one of the best blogs on the net. I will recommend this web site!
Your life will certainly never coincide hereafter!
Start feeling remarkable today by living the life that you’ve always desired!
Im grateful for the article post.Thanks Again. Awesome.
Major thanks for the article.Really thank you! Much obliged.
Good luck to you and your effort, dear teacher. Thank you.
Good luck to you and your effort, dear teacher. Thank you.
Terrific to obtain one of the most out of your money. Remarkable!